Last week, the 41st World Veterinary Association Congress was held in Tokyo. With attendance of 4,400 people from around the world, it was a very successful event. Their Majesties the Emperor and Empress also attended, joining us in listening to the keynote speech on highly pathogenic avian influenza virus strain mutations.
I also had the opportunities to reconnect with many veterinary friends, including former president Dr. Johnson Chiang, and learn the updated academic and clinical veterinary medicine from many splendid veterinary scientists and practitioners.
My tie was a gift from the congress held in Yokohama 31 years ago, and I still cherish it.
WVC2026
I attended the Western Veterinary Conference (WVC) in Las Vegas and listened to BCI’s 2026 Animal Health Industry Overview.
I strongly feel that both the animal health and veterinary industries in Japan are at a turning point, so I wanted to find any clues that could lead to a solution there.
I also had the opportunity to try out Zoox, a fully driverless vehicle offered by a subsidiary of Amazon. I was deeply impressed that a world where both human-driven and driverless vehicles already exist in Las Vegas.
AHRMS Telemed® Vet Prescription Platform Featured in S&P Connect / S&P Global Animal Health
AHRMS Inc. is pleased to announce that our patented veterinary prescription platform Telemed® System was featured by S&P Connect / S&P Global Animal Health, a leading global resource for animal health industry intelligence.
This feature highlights Telemed® as an innovative model designed to create a fair, transparent and sustainable distribution structure for veterinarians, wholesalers and pet owners — addressing challenges such as pharmaceutical inventory risks, staff shortages, long wait times, and the growing shift toward online distribution.
■ Comment from Yuki Ujimasa, CEO / President – AHRMS
“This publication is not only an honor, but also a gateway to international collaboration. Our Telemed® is more than a patent — it is infrastructure designed to bring fairness, transparency and continuity to veterinary medicine.
(Source: S&P Connect)
We look forward to collaborating with partners worldwide to help reshape the future of animal pharmaceutical distribution.”
Partnership Opportunities
AHRMS is now open to discussions regarding
global collaboration, licensing frameworks (exclusive / non-exclusive), and regional alliances.
If you want to read the full article and/or have any inquiries, please contact us thorough https://ahrms.jp/wp/inquiry/
Inspiring Young Veterinary Professionals: Dr. Ujimasa Delivers a Special Lecture at NVLU
On November 19th, Dr. Ujimasa had the privilege of delivering a lecture to second-year students in the Department of Veterinary Health and Nursing at the Nippon Veterinary and Life Science University. We are deeply grateful for this honorable opportunity.
Upon graduation, these students will take the national examination to become certified veterinary nurses. Approximately 60–70% are expected to work in veterinary hospitals throughout Japan, while an additional 10–20% will pursue careers in animal health–related companies.
At around 20 years of age, these students stand at the very beginning of their professional journey. Dr. Ujimasa encouraged them to embrace new challenges—whether in academic study or personal pursuits—with the mindset that “even if you fail, your life will not be taken.” This message reflects his belief in the value of bold curiosity and resilience for the future leaders of the animal health industry.

Crecon’s 58th animal health seminar

On October 8, I had the honor of speaking at the 58th Animal Health Seminar organized by Crecon Research & Consulting, where I gave a presentation to 144 participants from 43 animal health companies.
This was my sixth lecture in Crecon’s long-running seminar series, and the title of my talk was:
“Discounts can be Poison – Strategy is the Real Cure”.
More than ten years ago, the late Ron Brakke of Brakke Consulting, Inc. (BCI) once remarked during his visit to Japan that “the Japanese animal medicine distribution system is about 20 years behind the U.S.”
I sometimes imagine what Ron would say if he could see Japan’s situation today — how far we may have come, and how far we still have to go.
Yet, even now, I cannot help but feel the gap that remains between Japan and the United States.
That is why I continue to raise my voice, sound the alarm, and urge awareness of the challenges ahead.

Panel Discussion at 3rd Interpet Osaka
I participated in the 3rd Interpet Osaka as one of the panelists for the panel discussion titled “Future outlook for the Japanese pet industry” on June 13th, 2025.
Thank you to the many friends and acquaintances who came to support me.
I was harsh on both original and generic manufacturers of animal medicines, but I would appreciate it if they could provide unbiased and accurate information, otherwise wholesalers and veterinarians will spread misinformation to pet owners.
Facilitator Chairman Dr. Koshimura also said, “Currently, senior dogs and cats account for more than half of the total, and newborn puppies and kittens account for less than 4%, so a significant decrease in the number of pets kept is inevitable. In 7-8 years, veterinary clinics will begin to be eliminated. Can anyone give their opinion on this?” He asked all the panelists and I answered since I was a representative of veterinarians there.
My biggest concern is that owners will stop visiting veterinary clinics due to rising veterinary medical costs, which is actually happening in the United States and other countries.

keynote speech at the 56th general meeting of the Japan Animal Drug Instrument Distributor Association
On May 22nd, I had the opportunity to give a keynote speech entitled “Japan in the World – Trends in Animal Medicines” at the 56th regular general meeting of the Japan Animal Drug Instrument Distributor Association.
Some people told me that they attended just for this, and I would like to express my sincere gratitude for the opportunity.

Japan in the World – Japan’s Animal Drug Market
At the request of the Japan Animal Drug Instrument Distributor Association, I ran 9 series of the above-titled document for two years.
I hope you will read the whole series from the following URL below by using ChatGPT as they are written in Japanese.
https://jadida.or.jp/supply_repo.html
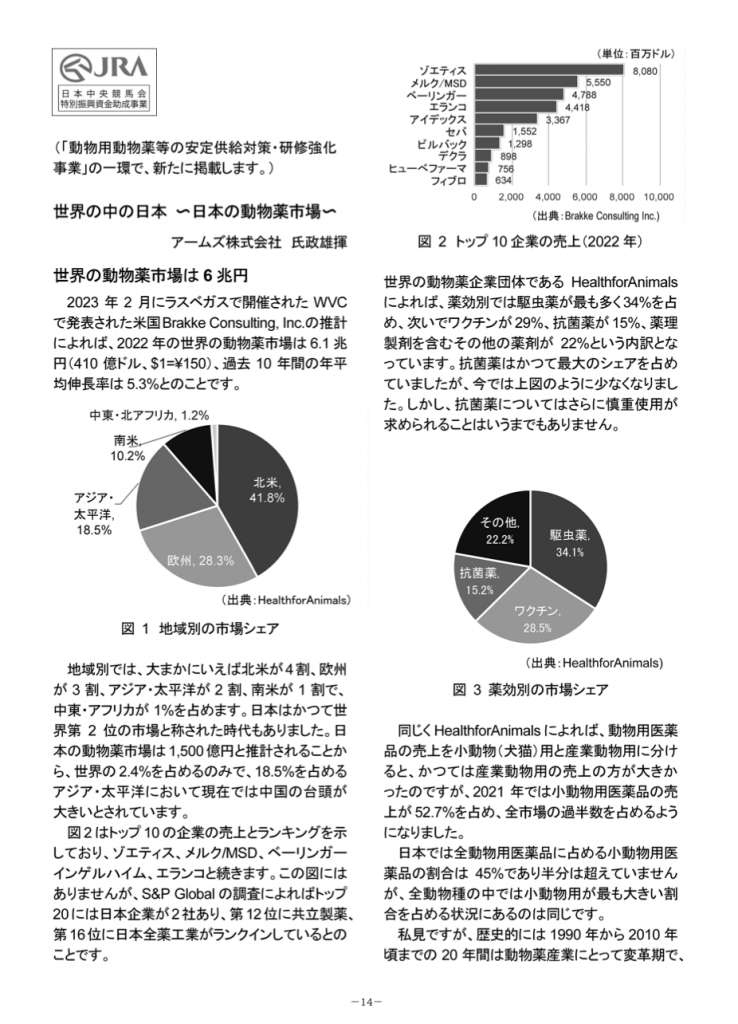
- The world’s animal drug market is 6 trillion yen
- MAFF is seriously cultivating the next generation of food industry
- Learn about the dynamism of the animal drug industry through corporate exhibits at WVC
- How will the world confront diseases for which no approved animal drugs have yet been approved?
- Is the fear of avian flu spreading from cows to humans in Japan unfounded?
- International cooperation on drug regulations and prohibited acts in horse racing
- Japanese animal drug companies should be more proactive in disseminating information overseas
- There are Japanese companies that specialize in research and development and are making a splash in the global market
- Unless Japan’s market value improves, we cannot expect the development of the animal drug industry or a stable supply
Issue of shortages of veterinary medicines was discussed at 29th AVC GA
I had the opportunity to speak at the 29th AVC General Assembly held in Utrecht, the Netherlands, about the issue of shortages of veterinary medicines, which has emerged as a post-COVID issue.
It was a very meaningful opportunity to discuss the issue not only with consultants from each country, but also with Dr. Schalansky of the EU (European Community) EMA (European Medicines Agency).


Stepping into the Japanese Animal Health Market
Would you like to grow your companion animal business outside the US but are not sure where and now? How about in one of the most valuable markets in the world, Japan? Brakke Consulting can help.

For the first time, Brakke Consulting will be presenting a seminar on taking the first step in Japan at the Western Veterinary Conference on Monday, February 19th from 9:30-10:30 am in the Acacia 1 room at the Four Seasons Hotel. This seminar will be presented by Yuki Ujimasa, DVM, MS, a Brakke senior consultant based in Tokyo, who has broad experience in the Japanese market. Yuki will focus on the steps needed to get your product approved by the regulatory authorities in Japan. This will be a great seminar for companies looking to expand their global business but need some help to get started.
Monday, February 19, 2024 at WVC
Four Seasons Hotel, Acacia Room









